WHY ELCAM
Your Source of Consistent Quality & Care
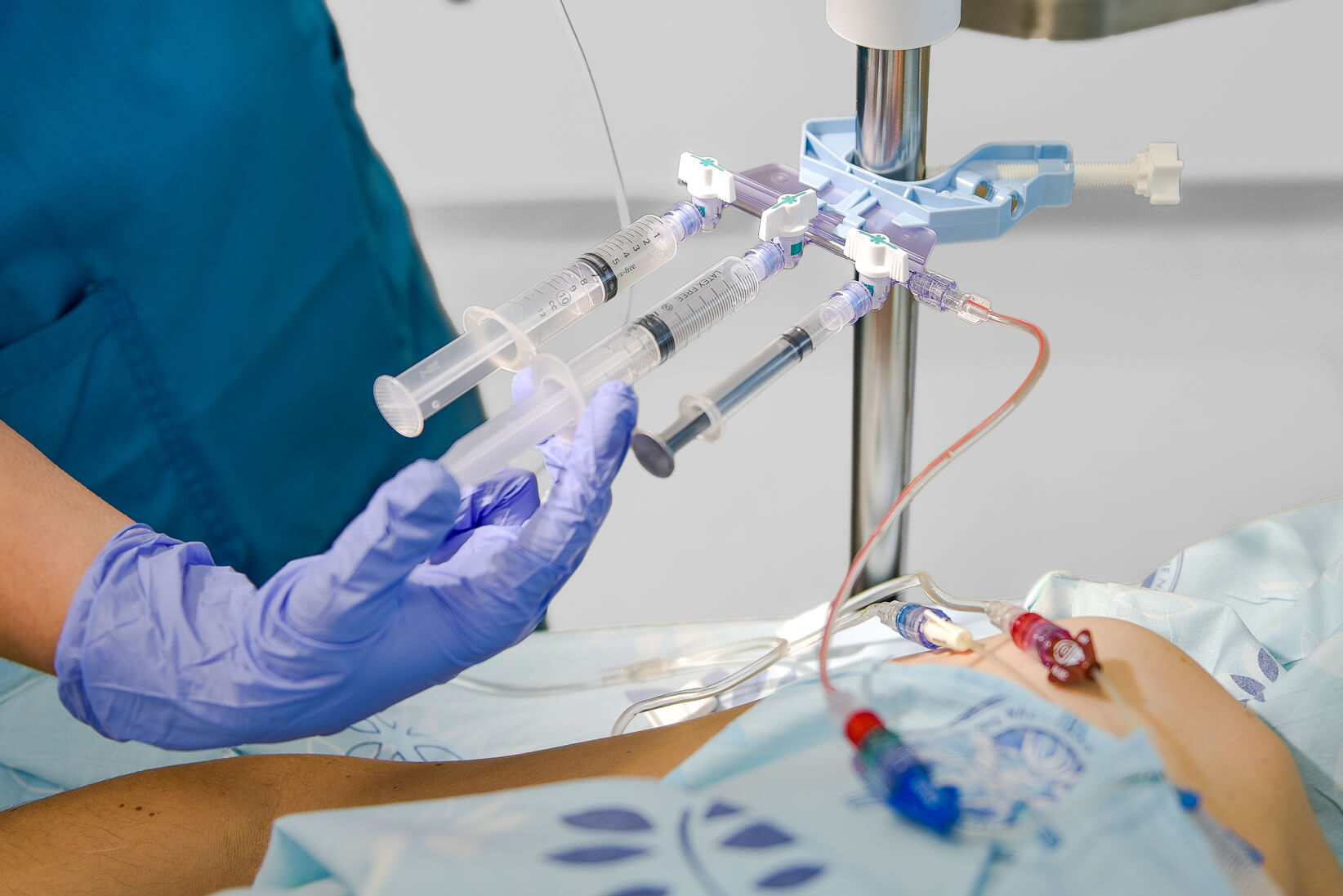
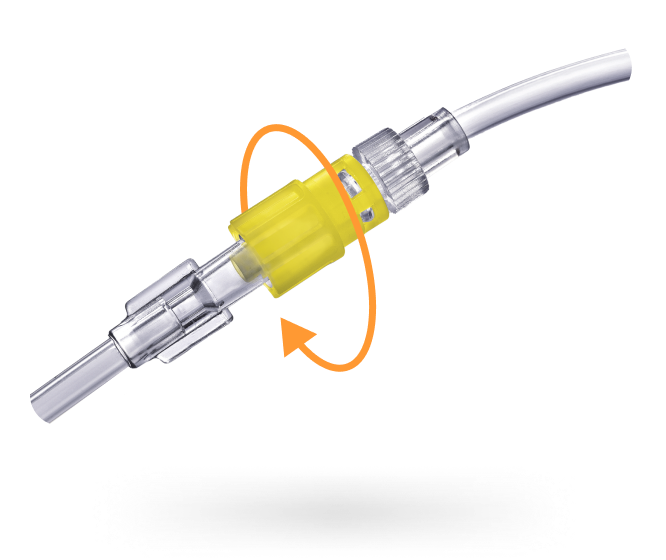
Elcam Medical is a leading force in medical device manufacturing. We specialize in OEM medical solutions, crafting disposable medical devices and accessories for the industry's top companies. Our focus is on innovative flow control solutions that contribute to safer and more effective fluid management in critical areas like Intensive Care, Anesthesia, Dialysis, Interventional Cardiology & Radiology, and Oncology. This expertise comes from our deep understanding of the medical and clinical environment, fostered by strong partnerships with our valued OEM customers.
Social responsibility
Sustainability isn't something we take lightly at Elcam, it's a core principle that guides our actions. We're dedicated to responsible corporate citizenship, achieving this through a strong focus on Environmental, Social, and Governance (ESG) practices. By implementing sustainable solutions, we foster stronger collaboration within our teams, build trust with customers and partners, and contribute positively to society as a whole. This commitment to ESG creates lasting value for everyone we touch.

Elcam’s Product
Development Process
Elcam goes beyond manufacturing. We offer comprehensive support throughout your product's lifecycle, from initial design and development to regulatory approval, launch, and beyond. Our engineering and regulatory expertise ensures a smooth journey.
Imagine the possibilities when you combine your innovative ideas with our proven methodology.